 , in which it can be seen that carbon
, in which it can be seen that carbon
Humic acids - the fraction of humic substances that is not soluble in water under acidic conditions (pH < 2) but is soluble at higher pH values. They can be extracted from soil by various reagents and which is insoluble in dilute acid. Humic acids are the major extractable component of soil humic substances. They are dark brown to black in color.Fulvic acids - the fraction of humic substances that is soluble in water under all pH conditions. They remains in solution after removal of humic acid by acidification. Fulvic acids are light yellow to yellow-brown in color.
Humin - the fraction of humic substances that is not soluble in water at any pH value and in alkali. Humins are black in color.
 , in which it can be seen that carbon
, in which it can be seen that carbon
| Humic acids of peaty sandy hydro earth soil | Humic acids of haplic phaeozems | Humic acids of podzolic soil |
|---|---|---|
  
   |
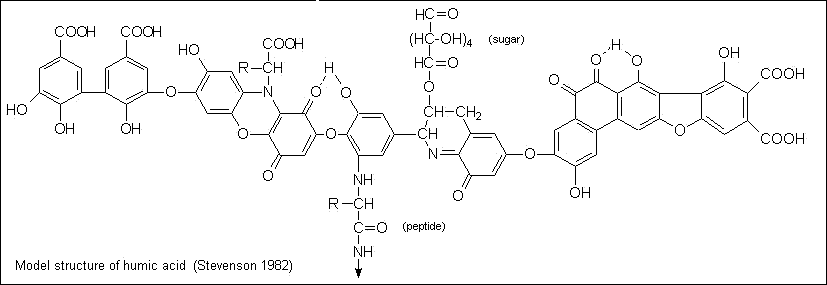
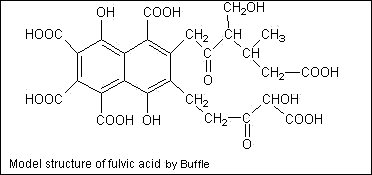
| Substances | % dry ash-free basis | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fulvic acids | 44 - 49 | 3,5 - 5,0 | 44 - 49 | Humic acids | 52 - 62 | 3,0 - 5,5 | 30 - 33 | Proteins | 50 - 55 | 6,5 - 7,3 | 19 - 24 | Lignin | 62 - 69 | 5,0 - 6,5 | 26 - 33 | |
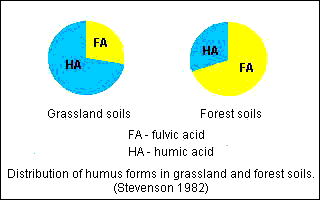
| Humic acid/ | Fulvic acid ratio Soil | Humic acid/ | Fulvic acid ratio Chernozem ordinary | Gray forest | Chernozem deep | Sod podzolic | Chestnut dark | Tundra | |
|---|