Signifcance of carbohydrates
The major groups of carbohydrates
- 1. Monosaccharides, which are aldehyde and ketone
derivatives of the higher polyhydric alcohols.
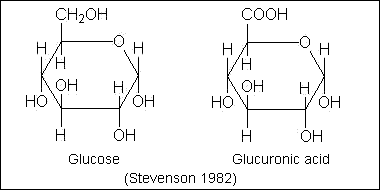
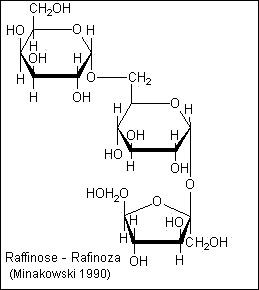
- 2.Oligosaccharides, a large group of polymeric
carbohydrates consisting of a relatively few monosaccharide units.
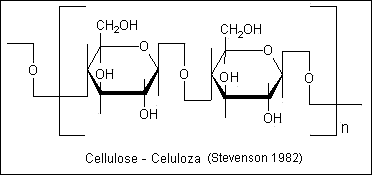
- 3.Polysaccharides- contain many monomeric
units (8 or more)
The carbohydrates material in soil occurs as:
2. complex polysaccharides
3. polymeric molecules of various sizes and shapes which are so strongly attached to clay and/or humic colloids.
The cotribution of individual sugar types to soil organic matter
(Stevenson 1982)
| Sugar | % of organic matter |
|---|---|
| Amino sugars | |
| Uronic acids | |
| Hexose sugars | |
| Pentose sugars | |
| Cellulose | |
| Others | |
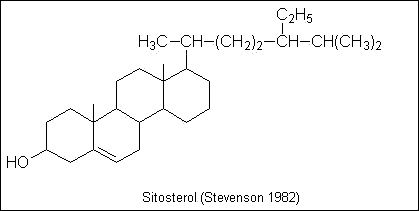
Soil lipids
Amino acids
- As free amino acids
- in the soil solution
- in soil micropores
- As amino acids, peptides or proteins bound to clay minerals
- on external surfaces
- on internal surfaces
- As amino acids, peptides or proteins bound to humic colloids
- H-bonding and van der Waals' forces
- in covalent linkage as quinoid-amino acid complexes
- As mucoproteins
- As a muramic acid